Services
Our supply chain expertise lets you offer the right product with the right quality, delivered to the right place and at the right cost.
Bushu Pharma's supply chain is built to offer the right product with the right quality, delivered to the right place and at the right cost. Our commitment to quality guides our decision making with the goal to provide a secure supply chain for our customers.
We strive to regularly enhance our supply chain organization to meet the increasing complexity driven by our clients’ product portfolios. Supply chain operations are integrated into our business across multiple sites and personnel required for forecasting, manufacturing, inspection, packaging and distribution.
From the point of manufacture through to local delivery, Bushu Pharma’s GMP know-how backs our SCM solutions, allowing you to control your supply chains with confidence. Our service will allow you to be able to comply with all traceability regulations, meet delivery commitments, and efficiently manage recalls.
-
Demand Planning
To support clients and to fulfil their demands and orders, we execute each function of the manufacturing process with close management of forecast, inventory, material control, vendor oversight, regular/urgent order, logistics, delivery, and lead-time oversight.
Another key factor is the ability to respond to a wide range of order sizes. With two large manufacturing and storage facilities in the greater Tokyo area, we provide assured capacity to meet the demand of service targets, stock and costs. -
Performance &
ReviewWe establish our culture of sustainable performance within our supply chain operation system, consistently checking and reviewing progress to ensure on-time delivery in all cases.
-
Distribution & GMP Certification
Bushu Pharma breathes GMP Manufacturing. It is what defines us and what we offer to each and every one of our customers.
Bushu Pharma offers its customers the highest level of expertise by emphasizing GMP Manufacturing. In addition to the three major GMP in Japan, the United States, and Europe, Bushu holds GMP certifications from countries across Southeast Asia, Central and South America, the Middle East, Africa, Canada and Australia.
We adhere to ICH*1, PIC/S*2, and practice GMP and GDP.
*1 ICH: International Council for Harmonization of Technical Requirements for Pharmaceuticals for Human Use
*2 PIC/S: Pharmaceutical Inspection Convention and Pharmaceutical Inspection Co-operation Scheme
This commitment permeates each of the four key deliverables Bushu promises its customers:
-
Latest Technology
-
Quality Assurance
-
Delivery Management
-
Cost Management
Through our commitment to GMP manufacturing, we strive to create efficiency in distribution structures and platform networks following GDP.
-
-
Engineering
We define, establish, control and improve the process flows of production, information and financial with modern digital technology.
-
Packaging and
ArtworkFrom the sourcing of suitable materials, to the design of packaging that can preserve the shelf life of your product, Bushu Pharma will consult closely with you to help bring your product to market.
Bushu Pharma's packaging service is able to handle press through packaging (PTP), bottle packaging, anti-counterfeiting products, serialization, bar code, and minimizing carton box size. Bushu Pharma also provides solutions from design to order label, packaging inserts, carton boxes, and tamper evidence seal for packaging materials.
Vendor Management
Bushu Pharma manages packaging and distribution suppliers in the quality agreement process, quality audits, packaging material specification, and incoming tests.
Artwork Management
Bushu Pharma is able to expertly handle packaging labeling (artwork) development and changes through the following steps:
- Draft artwork design
- Finalize artwork (approval)
- Procure packaging materials after artwork approval
- Acceptance test of packaging materials
-
Key Partnerships
To be the best, you have to work with the best. Hardly any company in today's global economy can excel without the support of a vast network of key suppliers and vendors, which is This is what we strive to do in order to create a reliable one-stop service - from manufacturing to packaging and for your business expansion needs.
We offer consulting services for business development / business model creation. We are experts in Japan market made possible by relationships with reliable vendors that allow us to create custom solutions for our customers.
-
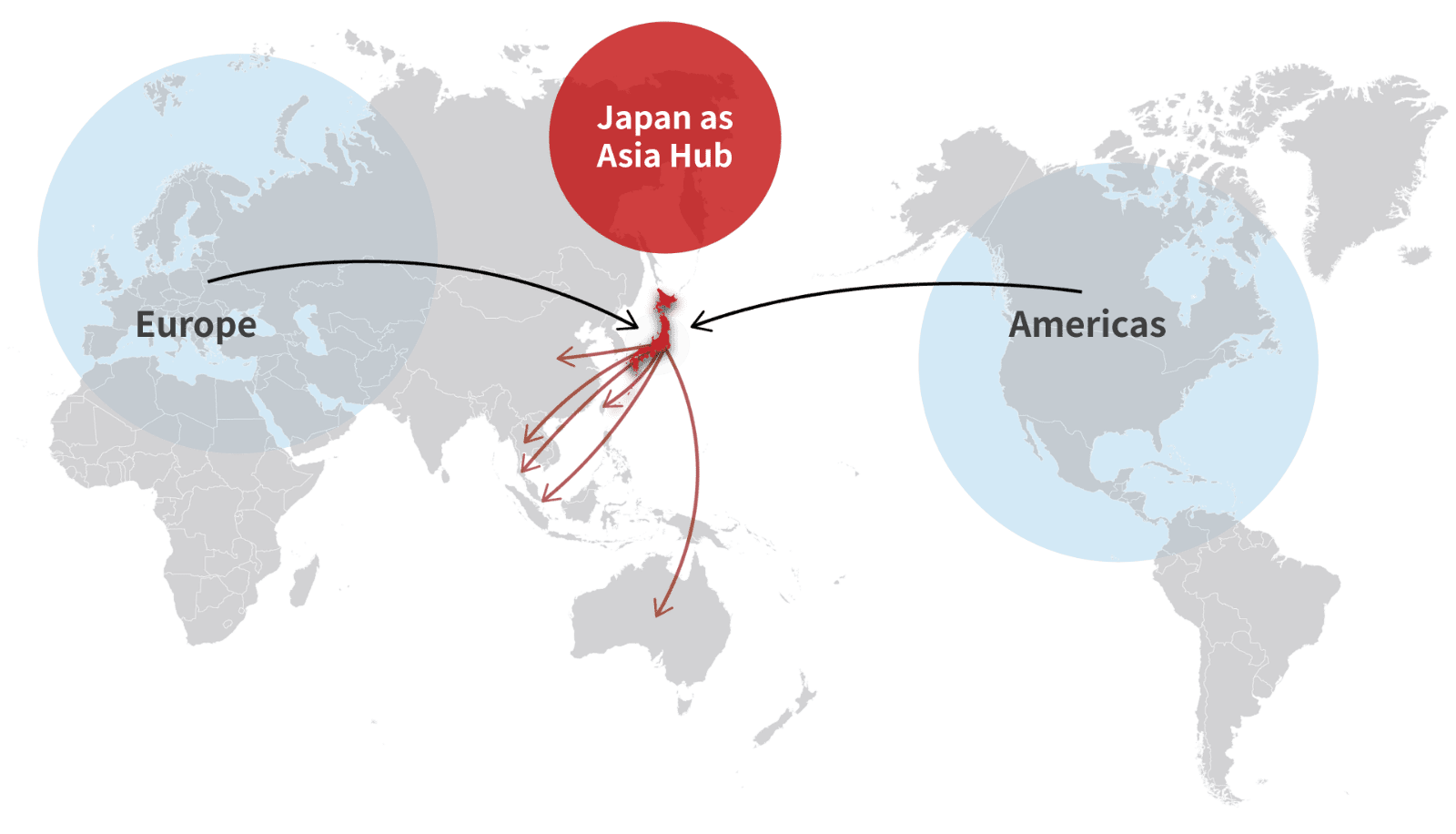
GATEWAY to ASIA®
Bushu Pharma's "GATEWAY to ASIA®" service supports customers in their desire to launch new products in Japan and across the larger Asian region.
Our GATEWAY to ASIA® service lets international pharmaceutical companies import bulk products into Japan for quality inspection, labeling, packaging and distribution throughout Japan and other Asian countries. The service lets you focus on your core businesses and avoid having to deal with the difficult process of managing lead-time, temperature, security, inventory, packaging and urgent order handling across multiple markets.
Collectively, our services offer reduction of lead time from purchase order to shipping, financial and physical flow, oversight of inventory management for Asian markets, forecast review, and planning and operations based in GMP expertise.
Why choose Bushu Pharma for your ‘Asia Hub’ and global expansion partner?
(Background) Japan is the world's third-largest consumer of pharmaceuticals, following the United States and China. To address the internationalization of the pharmaceutical supply chain, harmonization efforts are being conducted within the Pharmacopoeial Discussion Group (PDG), which consists of the Japanese Pharmacopoeia (JP), the United States Pharmacopeia (USP), and the European Pharmacopoeia (Ph. Eur.). Additionally, the Ministry of Health, Labour and Welfare and the Pharmaceuticals and Medical Devices Agency (PMDA) aim to reference the JP in the Asian region.
Given the background mentioned above, by utilizing our services, Bushu Pharma can provide the following support.
- · Japan ranks 3rd globally after the US and China in the world‘s drug cost country rankings.
- · International promotion of Japanese Pharmacopoeia (JP) in line with globalization of supply chain for pharmaceuticals. For example, harmonization of test methods and monographs is conducted by Pharmacopoeial Discussion Group (PDG) which consists of JP, USP, and EP. In addition, utilization of JP as a reference pharmacopoeia in Asian region is aimed by the MHLW(Ministry of Health, Labor and Welfare) and Pharmaceuticals and Medical Devices Agency of Japan./li>
- · Bilingual project management, with close communication from project kick-off to actual product launch
- · 56 GMP certifications and high skill for technical transfer
- · Ability to advise on packaging specifications by country and product specification
- · Long track-record of skilled vendor management for packaging materials
- · Familiarity with pre-approval inspection (PAI) and flexible planning for process operation
- · Temperature-controlled warehouse storage of finished products
- · Prompt and economical product delivery to wholesalers or distributors
- · Near flawless inspection history by foreign authorities
-
Global Expansion
SupportUsing the production capacity of our five factories in Japan, Bushu Pharma supports both Japanese and global pharmaceutical companies looking to launch their products in new markets.
We closely communicate with you from project kick-off to actual product launch, we will ensure we are the best partner for your product development.
Working with Bushu Pharma for your product launch lets you draw on our GMP know-how. We have GMP certifications for Japan, the US, Europe, and other countries such as of South-East Asia, Central & South America, the Middle East and Africa, and we can manufacture clinical samples in addition to pharmaceuticals.
With concern over containments never higher, let Bushu Pharma and our Japan High Quality manufacturing system help you reach new markets around the world.。
-
Cold Chain
LogisticsMany pharmaceutical and Biological products today are extremely sensitive to the conditions in which they are packaged, shipped and stored, with temperature deviations affecting the quality of product delivered. Bushu Pharma is skilled in the management of these factors and will ensure your products are handled with the specialist care and the attention that they require.
We are able to help improve the packaging and storing process which must be validated to ensure that there is no negative impact to the safety, efficacy or quality of the drug.
Through our Cold Chain logistics service, we enable the market expansion for temperature-controlled bio-pharmaceuticals (maintained at 2℃ to 8℃) to the Japan market, as well as to countries across Asia and beyond.
We store and transport pharmaceuticals in line with each product's specific storage and security requirements, maintaining cold chain integrity throughout each step of the delivery process.
-
1. Cold Warehouse Management

-
2. Packaging Exposure Time Management

-
3. Cold Logistic Management

We handle storage management not only within the temperature range of 2-8°C but also collaborate with customers to accommodate storage at extremely low temperatures such as -20°C, -40°C, and -80°C.
-
-
Distribution and
Quality AssuranceBushu Pharma constantly strives to be flexible in the way we work with our customers, aiming to help each and every customer reach their business goals.
Distribution is an important component of the integrated supply-chain management of pharmaceutical products.
Bushu Pharma ensures the quality and identity of pharmaceutical products during all aspects of the distribution process. These include procurement, purchasing, storage, distribution, transportation, re-packaging, re-labeling, documentation and record-keeping practices.
When the distribution chain is faced with the manufacturing steps such as repackaging and relabeling, we strive to apply the principles of GMP to these processes.
Bush Pharma will always do its best to make every activity of pharmaceutical distribution in accordance with the principles of GMP, Good Storage Practice (GSP) and Good Distribution Practice (GDP).
Bushu Pharma constantly strives to be flexible in the way we work with our customers, aiming to help each and every customer reach their business goals.
Even at low volume, we can also manage operations with manual visual inspection, operate semi-auto lines, provide manual packaging, and deliver final products directly to the destination designated by each customer.
Additionally we manage urgent delivery and priority bookings (which is especially relevant for new product manufacturing by contracting formulation), packaging and QC testing. We make this possible by taking advantage of a shift-based working structure, whereby our staff work in three staggered daily shifts, as well as during special campaign operations.
CONTACT
As a company specializing in contract manufacturing of pharmaceutical drugs, we offer a wide range of contract services tailored to clients’ needs based on our extensive experience and know-how accumulated over many years.