Services
We offer contract services that support the manufacture of commercial products and clinical trial supplies in solid dosage formulations, solid dosage-drug packages, and injectable preparation packages in compliance with requirements of our customers by implementing our expertise accumulated over 25 years of experience as a CDMO.
We accept contracts for the manufacture of pharmaceutical products and clinical samples of solid dosages and injectables - offering an unprecedented breadth of expertise in technical transfer and analytical method development.
Supported by strong manufacturing facilities and processes, from start to certification, Bushu Pharma always strives to improve its ability to offer clients the best solutions for material procurement, technical transfer, production, packaging, logistics, and distribution - in Japan and across Asia.
We value close communication with our customers, and manufacture products for clinical studies and commercial sales in line with the needs of customers as well as those of patients to support the efforts of our customers in achieving success of their projects.
We also offer services of supplying products with standards of quality required for commercial sales and with stability of quality maintained while passing through multiple distribution channels, in line with global pharmaceutical regulations.
-
Solid Dosage Formulation
Bushu Pharma provides contract manufacturing services for the production of granules, powders, tablets and capsules.
-
Dosage forms
- · Granules, Fine granules, and Powder
- · Plain tablets
- · Film-coated tablets
- · Sugar-coated tablets
- · Multi-layer tablets
- · Orally disintegrating tablets
- · Dry coated tablets
- · Hard capsules
- · Soft capsules (manufacture of solutions for internal use)
-
Manufacturing features of solid dosage forms
- · Explosion proof facilities
- · Coating of fine particles
- · External lubrication system of a tablet press
- · Capsule filling unit
- · Small scale to large scale manufacturing
- · Crushing of bulk drugs
- · OSDrC technology (dry-coated tableting)
-
-
Injectables
Bushu Pharma provides contract manufacturing of injectables, vials, lyophilized vial and pre-filled syringes
Inspection and Packaging Injectables
We inspect and package filled vials, syringes, and ampoules manufactured overseas. Our large refrigerated warehouse enables us to handle clinical samples and pharmaceutical products that require refrigeration.
Ensuring your product with no defects is essential to customers’ success in any markets. Bushu Pharma has strengthened its Inspection and analysis. We have the know-how, skillful inspectors, analysts and equipment to expertly conduct inspection and analysis of your product – checking for visual defects of both the product and packaging.
This function is a core part of our Bushu Pharma for injectables. Three key areas that are focused on to ensure all products meet quality requirements for the Japan market are:
- Inspection of the cosmetic quality of bulk drug products
- Inspection of the cosmetic quality of packaged products
- Analysis for the presence of foreign materials
In order to meet market expectations for quality control in each of these areas, we employ the general following practices:
- Injectable products:
- Products in vials and syringes are rigorously inspected following the filling process to detect and remove any microparticles in solution such as glass and fibers, to meet the specified regulatory requirements for distribution within Japanese markets and overseas.
- Finished products:
- The cosmetic quality of finished drug products can be inspected by well-trained operators who remove defects – such as dents or scratched carton boxes.
- Foreign materials:
- Once foreign particulate matter is detected, we perform analyses using a range of instruments to identify root cause(s) of entry and propose improvements of the manufacturing process.
Factory personnel responsible for the inspections listed above are skill-certified personnel. It is obligatory for them to undergo annual skill testing for certification renewal.
To support quality control in terms of foreign materials, we make quick response to market complaint and foreign matters found in manufacturing process. Both non-destructive and destructive analysis are available. Our knowledge and experiences of manufacturing is the practical approach the foreign materials from the results of analysis. As our representative scheme, we make Microscopic observation, Element analysis by X-ray Microscope, Identification by Raman, and Identification by FT-IR.
-
High Potency Product Formulation
We manufacture formulation of high potency products ranging from clinical samples to pharmaceuticals
Our manufacturing environment is based on hazard assessment, exposure assessment, and implementing strict residue management.
In addition, we monitor the environment and verify the containment by measuring the exposure to workers. We target APIs with an OEL (Occupational Exposure Limit) of 1μg/m³ or less. The production scale is mainly 5 to 15 kg, and the dosage forms are granules, uncoated tablets, and coated tablets.
Based on the safety information, we evaluate the APIs and decide whether we can handle them or not, taking into account the dosage form and the manufacturing process.

Main Equipment for High Potency Product Formulation
High speed agitation granulator / Fluidized bed granulator / Particle sizing equipment / Tableting machine / Packing machine
High Potency Tablet Dosage Form
-
OEL
0.15-1μg/m³(principle)
-
Scale
5kg-15kg
-
Dosage
formGranule/ Plain tablets/
Coasted tablets
-
Production Equipment
- Closed system
- Glove box
- Flexible enclosure
- Isolation flap
- Local exhaust ventilation system
-
Production Area
- All fresh air-conditioning system
- Monitoring system
- Mist shower
- Independent dedicated air-conditioning system
- Dedicated purified water generation system
-
Rigorous Operating Policy
- Cleaning validation
- Dedicated equipment and instruments
- Entire cleaning procedure for production room/auxiliary chamber
- Prevention of cross-contamination
- Monitoring of exposure/leakage
- Disposal of cleaning tools and clothes
-
-
Packaging
We provide primary to final packaging services for a wide range of product items.
-
Solid Form
Drug Products
- · Granules
- · Tablets
- · Capsules
- · Dry Syrup
Primary / Secondary Packaging
- · Blister
- · Stick Package
- · Bottle
-
Injectable Form
Dosage forms
- · Vials (liquid and lyophilized products)
- · Pre-filled syringe
Inspection Assembling
- · Vials
- · Pre-filled syringe
- · Auto Injector
Secondary Packaging
- · Vials
- · Pre-filled syringe
- · Auto Injector
-
Medical Device
Secondary Packaging
- · Inspection of medical devices
- · Packaging of drug-device combination products
Regenerative Medicine
Secondary Packaging
PTP and bottle packaging
- · We are equipped with filling machines suitable for packaging of two-sided aluminum medical blister packs.
Packaging Line Highlights
- · Automatic line
- · In-line visual inspection cameras
- · Packaging under strict controlled conditions at low temperature (2-8°C)
Packaging and Artwork
From the sourcing of suitable materials to the design of packaging that can preserve the shelf life of your product, Bushu Pharma will consult closely with you to help bring your product to market.
It is possible for us to print on PTP packages, bottle containers, anti-counterfeiting packages, GS1 Bar Code, etc. We offer a variety of technical solutions, for example package design, such as minimizing the size of individual boxes, and support for artwork designing.
Vendor Management
Bushu Pharma manages packaging and distribution suppliers in the quality agreement process, quality audits, packaging material specification, and incoming tests.
Artwork Management
Bushu Pharma can expertly handle packaging labeling (artwork) development and changes through the following steps:
- Draft artwork design
- Finalize artwork (approval)
- Procure packaging materials after artwork approval
- Acceptance test of packaging materials
-
-
Clinical Trial Supplies (CTS)
We have a wealth of experience in contract manufacturing such that we are able to produce clinical trial materials to the specifications of our clients. In addition to manufacturing placebos, we can provide packaging for double-blind testing.
Bushu Pharma has accommodated an increasing demand for clinical samples each year. We cover contracts from shallow, small-scale and early phases to large-scale and final phases.
Bushu Pharma has a well trained Clinical Trials Team in our packaging department, providing the following services:
- · Package engineering
- · Package material management
- · QC test
- · GMP release
- · Product delivery
-
Analysis
Under our GMP system based on extensive experience with inspections in Japan and overseas, we are able to conduct tests on a wide range of pharmaceutical products from low-molecular weight drugs to high-molecular weight drugs. In particular, we have the technologies and facilities to handle the testing of bio-pharmaceuticals such as antibody drugs, and we are able to provide testing using cultured cells (cell-based assay) that require sophisticated techniques.
Under our GMP system based on extensive experience with inspections in Japan and overseas, we are able to conduct tests on a wide range of pharmaceutical products from low-molecular weight drugs to high-molecular weight drugs. In particular, we have the technologies and facilities to handle the testing of bio-pharmaceuticals such as antibody drugs, and we are able to provide testing using cultured cells (cell-based assay) that require sophisticated techniques.
We also have a laboratory for high potency substances and technologies to analyze foreign matter and are able to meet the varied analytical needs of our customers with regards to pharmaceutical products.
-
Medical Devices
At our Medical Device Centre, we manage the inspection and packaging of medical device combination products, drug delivery, and medical grade materials + devices.
For DDC (drug & device combination), Bushu Pharma manages the following operations:
- · Inspection of medical devices
- · Packaging of drug-device combination products
-
Animal Healthcare Products
We manage animal healthcare products and have manufacturing licenses for both animal healthcare products and animal healthcare biological products.
Our experts are able to respond to the needs associated with veterinary medicine. Industrial transfers can be made smoothly through the use of equipment which is similar to their existing manufacturing equipment. Our GMP laboratory can also carry out stability studies.
-
Technical Transfer of Analytical Methods
Bushu Pharma has a wide range of experience in technical transfer – from technical and quality control testing to certification and distribution.
We accept contracts for transfer of the manufacture of pharmaceutical products and clinical samples of solid dosages and injectables as well as related quality control tasks.
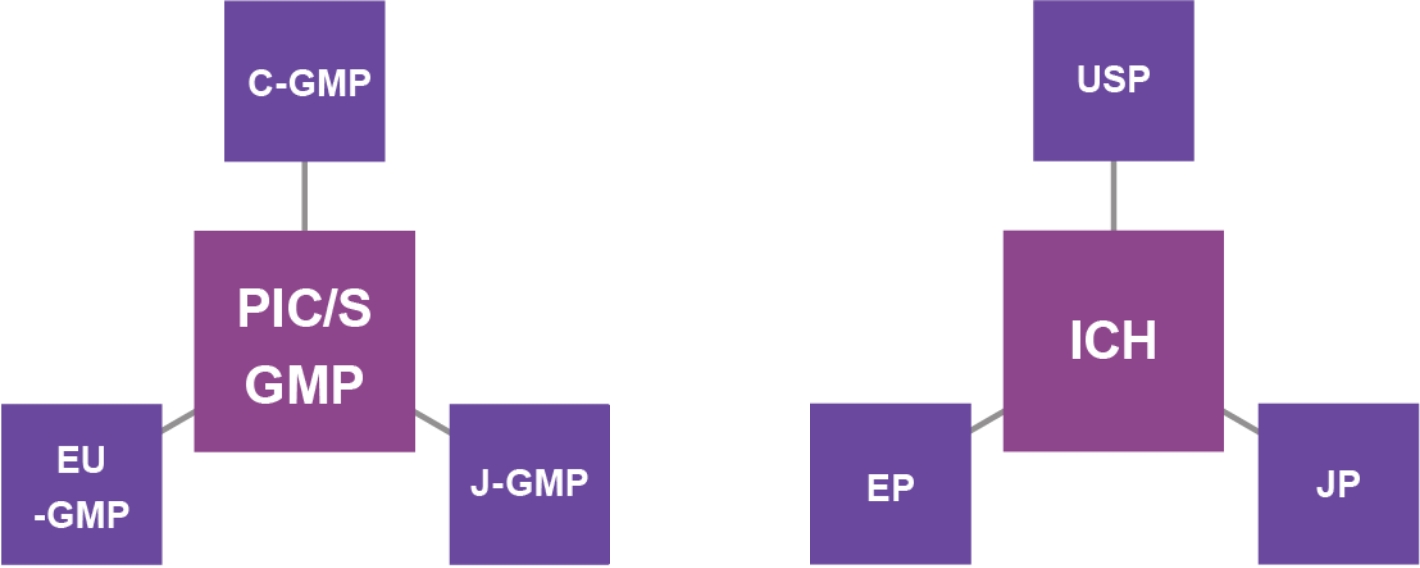
Our expertise in technical transfers has been developed over 20 years through QC Technical Transfer of Analytical Methods under USP, EP, JP and ICH.
CONTACT
As a company specializing in contract manufacturing of pharmaceutical drugs, we offer a wide range of contract services tailored to clients’ needs based on our extensive experience and know-how accumulated over many years.











