Quality Management System
Quality Management System
- Home
- Quality Management System
- Quality Management System
Quality Management System
Highlighted by the number of GMP certifications we have received over the years. Now operating as a CDMO for more than 25 years, we are proud that nearly half of our business generates from overseas customers. Whether in Japanese or English we are able to communicate in the language you speak.
-
Quality Assurance & Process
We have a comprehensive quality assurance management system that includes risk management procedures to ensure the highest quality and reliability of products.
Key Elements of our Quality Assurance Procedures
- A quality committee meeting held monthly to review the effectiveness of individual GMP systems. The Committee reviews the effectiveness and CAPA, the appropriateness of OOS, complaint management, and the status of the survey results of the contractor and public GMP.
- Established individual procedures (SOPs) and other documents for each GMP activities and GMP documents are managed by Parma Document, an electronic QA Document System that we have recently introduced.
- The data used for making decisions on developing, manufacturing, testing, delivering, and product monitoring at Bushu Pharma follow the principles of ALCOA+ (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring and Available)
- A wide variety of tests can be performed by QC.
LIMS (Laboratory Information Management System) is used in the QC Lab as well.
Note)As of October 2023, we are promoting the introduction of the Laboratory Information Management Systems (LIMS) to the Aizu factory.Going forward, Bushu Pharma remains dedicated to delivering the highest levels of Japanese craftsmanship and disciple in the manufacture, analysis, and distribution of pharmaceuticals and other medical products.
-
Inspections
Ensuring your product without defects is essential to your success in any market. With the expectation for defect-free product always in mind, Bushu Pharma has strengthened its Inspection and Analysis capabilities.
Solid Dosage products:
the cosmetic quality of bulk drug product can either be inspected by automated inspection machines or well-trained operators, and defects – such as foreign materials, dirty spots, chipped tablets, pigment agglomeration, etc. – can be removed accordingly.
Injectable products:
foreign materials in filled vials and syringes manufactured overseas can be inspected by highly-trained operators to remove defects – such as small glass fragments and fibers – to meet Japanese market requirements.
Finished products:
The cosmetic quality of finished drug products can be inspected by well-trained operators who remove defects – such as dents or scratched carton boxes.
To support quality control in terms of foreign materials, we make quick response to market complaint and foreign matters found in manufacturing process. Both non-destructive and destructive analysis are available. Our knowledge and experiences of manufacturing is the practical approach the foreign materials from the results of analysis. As our representative scheme, we make Microscopic observation, Element analysis by X-ray Microscope, Identification by Raman, and Identification by FT-IR.
-
Technical Transfer
Over 20 years, Bushu Pharma has developed a wealth of experience in technical & QC testing method transfer under USP, EP, JP and ICH protocols.
While technology transfers can oftentimes be characterized by a loss of quality or continuity from the product’s original purity, Bushu Pharma places the highest priority establishing a full understanding of the transfer at hand from our customers, taking into account the need to create speed and efficiency in the formulation process - but without the loss of quality.
For our existing customers as well as for companies considering a new manufacturer, you can count on us to handle your technical transfers and inspections from concept to commercialization with the utmost care.
Our technology transfer experts work with you for:
- · Site Transfer
- · Equipment Selection and Validation IQ, OQ and PQ
- · Existing Process Optimizations
- · Batch Record Development
- · Validation Protocol Preparation and Execution
- · SOP Development and writing
- · Alternate Raw Material Evaluations and Reformulations
- · In-Plant Troubleshooting and Investigations
- · Scale Up Assistance from feasibility to commercialization
Key features of our Inspection and Technical Transfer service
- · Fully supportable from early to late stage by SME team
- · Manage for each project and/or client by dedicated team consisting of SMEs
- · Assign SME, e.g. bioassay, biological, etc. for QC method transfer
- · Available bilingual approach, English and Japanese
- · Technical Services, PMO and Technical Operation Strategy are under COO in order to manage directly
-
Certifications
As a global contract manufacturer, GMP certification is key to being able to deliver results for our customers.
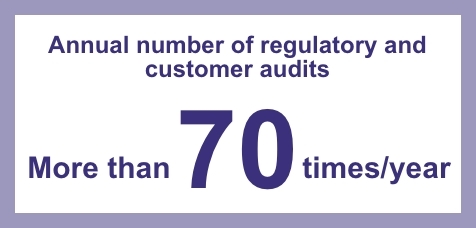
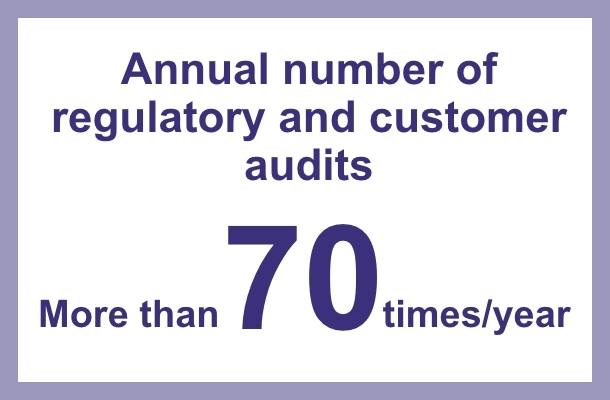
Our Kawagoe factory has acquired GMP certifications for Japan, the United States, Europe, Canada and Central & South America, while our Misato factory is GMP certified not only in Japan, the United States and Europe, but has also acquired GMP certifications from many countries including South-East Asia, Central and South America, the Middle East and Africa. We face regular client and regulatory audits each year and have been successfully audited by the FDA and other international authorities in recent years.
Three key areas that are focused on to ensure all products meet quality requirements for the Japan market are:
- · Inspection of the cosmetic quality of bulk drug products
- · Inspection of the cosmetic quality of packaged products
- · Analysis for the presence of foreign materials


CONTACT
As a company specializing in contract manufacturing of pharmaceutical drugs, we offer a wide range of contract services tailored to clients’ needs based on our extensive experience and know-how accumulated over many years.